- Easy and safe scale-up since your crystallizer consists of the same plate elements as used in our pilot plants
- Easy operation, since there is no crystal slurry handling and no filtration
- Our customers report high reliability and low operation costs
- Environmentally safe process due to inert gas blanketing and the absence of solvents
- Melt crystallization is an appealing option for your heat sensitive products, because of low operating temperatures around the melting point
- Customers are impressed by the simple operation and the high reliability of our crystallization technology
- Customers appreciate the high flexibility and wide operating range possible with static crystallization.
- A high yield can be achieved, even for viscous products.
Static crystallization
Win high yields with our static crystallization methods
- Organic and inorganic chemicals at high purities
- Monomers
- Carboxylic acids
- Aromatic isomers
- Tar chemicals
- Waxes
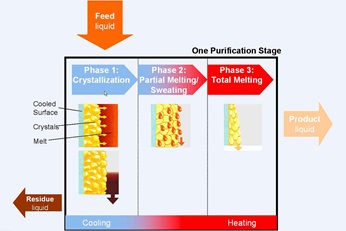
Static crystallization principle
Slow cooling of the heat transfer medium below the freezing point of the melt causes a structure of crystals to build up on the outer surface of the plates. Impurities are rejected from the growing crystals and are concentrated in the remaining melt. After the desired fraction has been crystallized, the remaining liquid phase is drained from the crystallizer. The purer crystalline layer remains adhered to the plates.Sweating or partial melting
This layer is further purified by sweating (partial melting), that is, by gently heating close to its melting point. Trapped and adherent melt, which contains the impurities, drains off. This partial melting step serves to further purify the crystalline product. Partial melting also rinses the equipment free of impure melt, preventing back-mixing and contamination of the final purified fraction.
Melting
After sweating, the purified crystal layer is totally melted and drained to the purified liquid product storage.
In general, crystallization is often the best alternative when distillation performs poorly, or if your product cannot be purified or separated by distillation due to close boiling points, or azeotropes between the components. In addition to static crystallization, our portfolio of melt crystallization technologies is completed by falling film and suspension crystallization. The chance that any of these work for your applications is high.
Contact our team below for your custom solution